Uncovering the Mechanism of Aggregation of Human Transthyretin
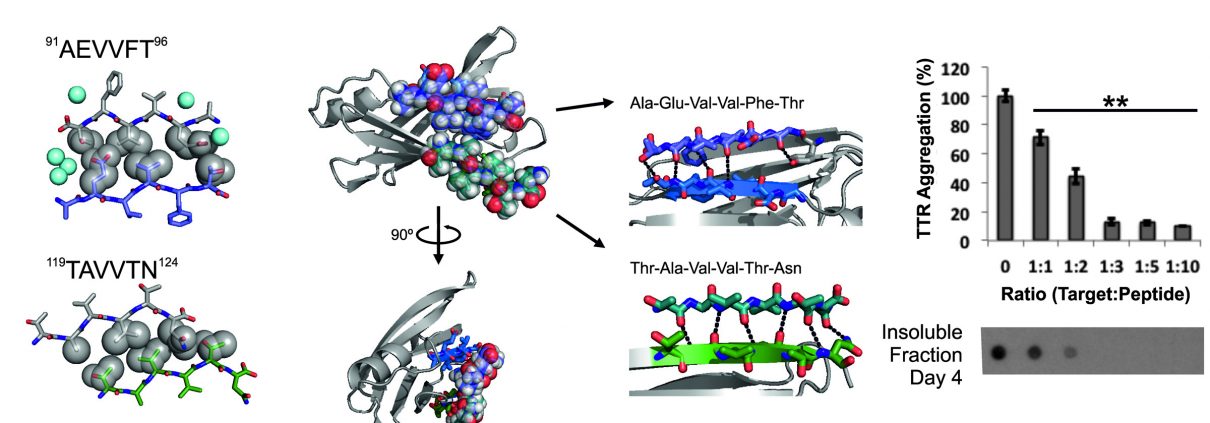
Published in the Journal of Biological Chemistry, the work of Dr. Lorena Saelices and coworkers under the supervision of Prof. David Eisenberg establishes a novel therapeutic strategy for transthyretin amyloidosis. The team identified two segments of transthyretin that are responsible for the protein aggregation and determined the structure of their fibrils. The fibril structures were used to design peptide inhibitors that block self-association of the two segments, and hinder transthyretin amyloid formation. The research published in December, 2015 opens a new approach to tackle the life-threatening transthyretin amyloidosis.
Abstract of the manuscript:
The tetrameric thyroxine-transport protein transthyretin (TTR) forms amyloid fibrils upon dissociation and monomer unfolding. The aggregation of transthyretin has been reported as the cause of the life-threatening transthyretin amyloidosis. The standard treatment of familial cases of TTR amyloidosis has been liver transplantation. Although aggregation-preventing strategies involving ligands are known, understanding the mechanism of TTR aggregation can lead to additional inhibition approaches. Several models of TTR amyloid fibrils have been proposed, but the segments that drive aggregation of the protein have remained unknown. Here we identify beta-strands F and H as necessary for TTR aggregation. Based on the crystal structures of these segments, we designed two non-natural peptide inhibitors that block aggregation. This work provides the first characterization of peptide inhibitors for TTR aggregation, establishing a novel therapeutic strategy