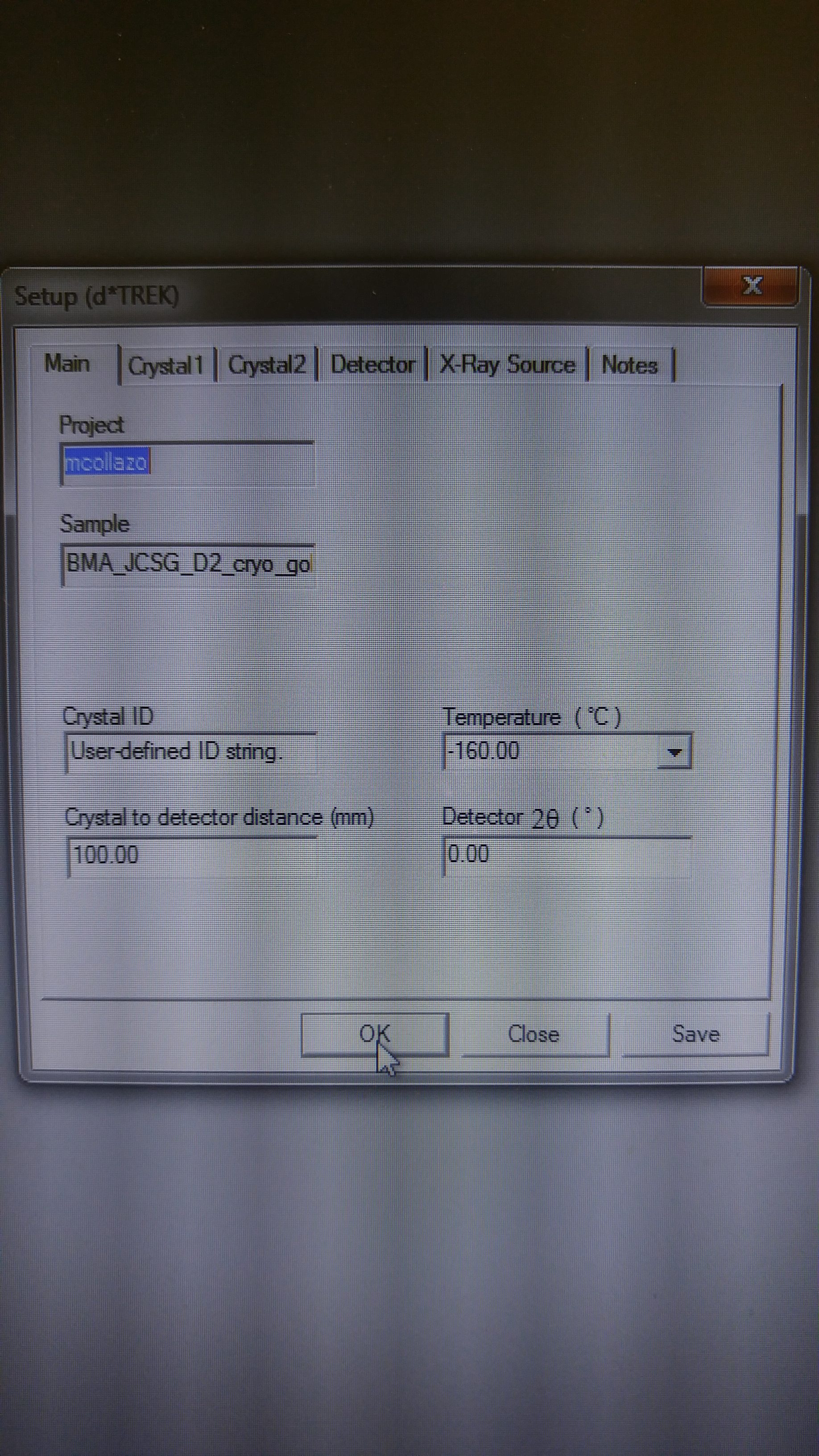
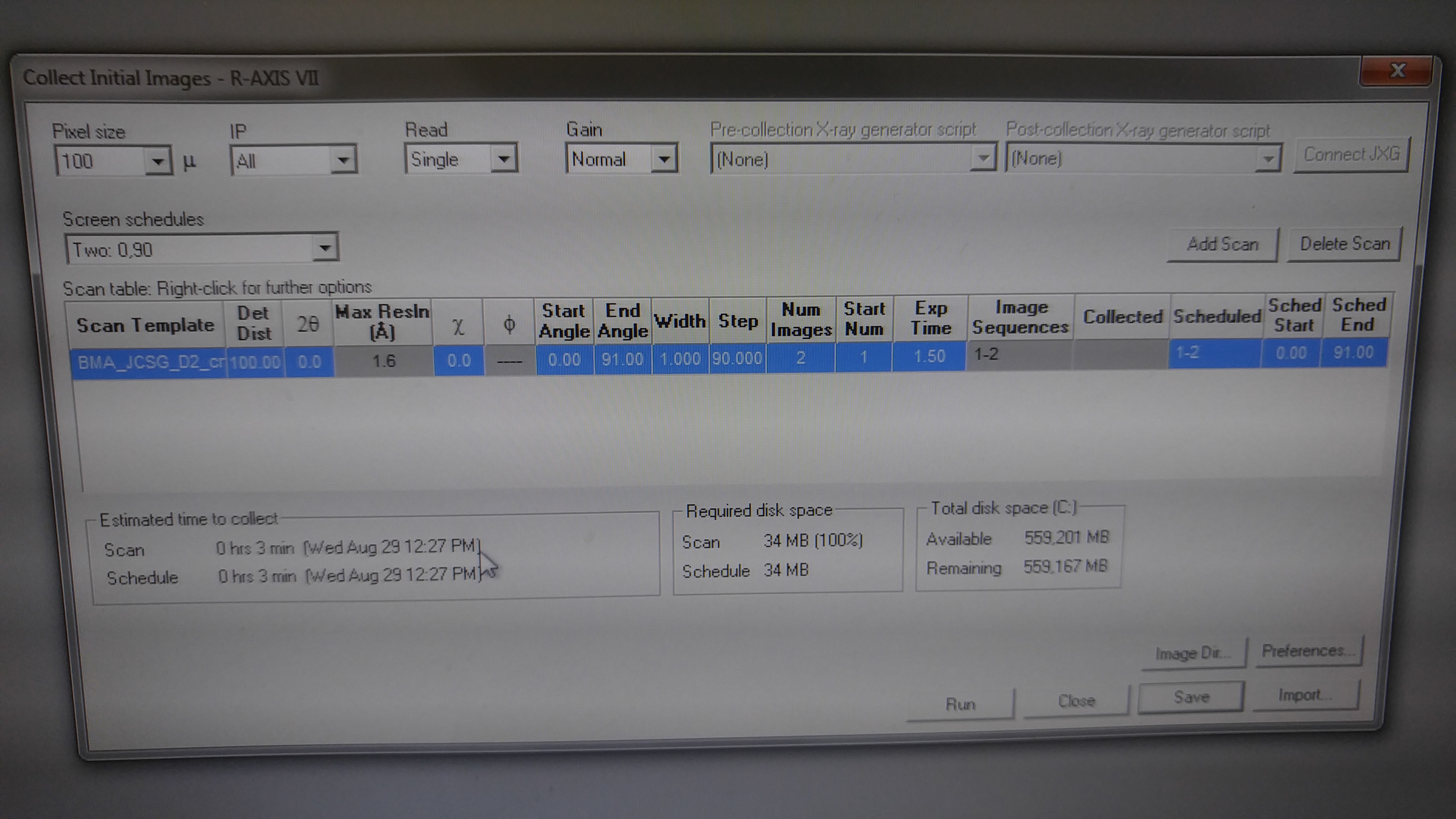
DOE INSTRUMENTATION CORE: X-RAY DIFFRACTOMETER
Tools/ Supplies:
-Magnetic Cryo-Wands -Pipettemen (0.5-10µL)
-Tweezers (crude) -Magnetic Crystal Loop Tips
-Scalpel -Glycerol (if cyro-protecting)
Access:
UNAUTHORIZED personnel in the x-ray lab will have their access revoked and will be reported to their principal investigator. DO NOT use the machine or enter the lab unless you are escorted by Duilio Cascio. You may enter the lab and use the machine ONLY IF three conditions are satisfied: (1) you are trained and authorized by Duilio Cascio to work independently. Secondary training is forbidden. (2) You complete EH&S diffraction safety training and give your certificate to Duilio Cascio. (3) You are given a key to the lab door.
DO NOT give your key and/or door code to another person even if they are authorized to work independently. Any violation will result in suspension of access cards for all parties involved. There are no exceptions.
Reserving a Time Slot:
-The Right-hand port (HTCR) on the right side of the board is suggested when shooting smaller crystals. HTCL has a larger beam diameter and may decrease the signal-to-noise ratio.
-Depending on the size and quality of your crystal sample, a longer period of time may be necessary for data collection. Be prudent when reserving a time slot.
-Consult with a staff member if you’re unsure about which of these will be best for your crystal.
-Write your name, day, and time you want to use the machine. This is not a guaranteed slot. You will have to wait if data is being collected when you arrive or the machine is being maintained. Be courteous to others and DO NOT shoot crystals if another user is signed up.
Fishing for Your Crystal:
-Select a loop of the correct size to match your crystal sample. Before opening your crystal tray. Use the microscope to verify the loop is undamaged and the correct size to catch your crystal. It is important that the loop tip be made of copper if you plan on collecting data for a long period of time. The copper tip resists freezing, but the others do not.
*It is important to verify this first to reduce the amount of time the crystal is exposed to air. Evaporation can often be detrimental and will reduce the quality of the data.
-Using the microscope, scan the wells to find your crystal.
*If you plan to cryo-protect your crystal, prepare a coverslip with a 2µL drop of glycerol and set this aside for later. Pipette an additional 4µL of mother liquor into the 2µL drop of glycerol. Glycerol resists the build-up of ice around your crystal sample and reduces the amount of noise in the diffraction pattern. Pipette an additional 4µL of mother liquor into the 2µL drop of glycerol.
-With a scalpel, carefully cut a perimeter around the well containing your crystal. Make generous cuts that over-lap at the corners.
-Use tweezers to remove the coverslip and place this under the microscope.
-Holding the cyro-wand as near to the loop as possible (without touching it), orient the tip so it lies flat (parallel) to the surface of the coverslip.
-Approaching at a 45° angle, gently nudge the crystal to dislodge it from the coverslip. Sliding in from beneath it and lifting the crystal is generally the best way to “catch” it.
-Try to be as precise as possible. This prevents unnecessary touching of the crystal which damages its integrity.
-In addition, do not hold the loop at less than 45° because this will cause the copper tip to act as a capillary and and draw your crystal out of the solution onto the copper base. If this happens the crystal is often lost.